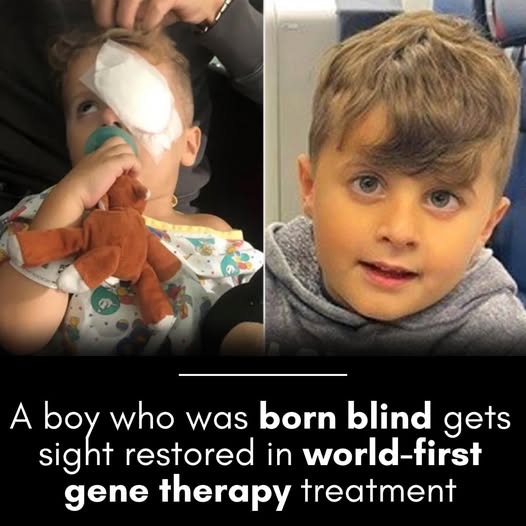
6-Year-Old Girl Sees After Life-Changing Eye Gene Therapy
A groundbreaking moment in pediatric ophthalmology — a 6-year-old girl born with a rare inherited retinal condition has experienced the miracle of sight for the first time following an innovative gene therapy procedure. This remarkable case represents not just a personal victory for one family, but a significant leap forward in how we approach childhood blindness caused by genetic disorders.
The child, diagnosed with Leber congenital amaurosis (LCA), a severe retinal degenerative disease that typically leads to near-total vision loss from infancy, underwent a targeted gene therapy that restored functional vision within weeks. Her story has captured the attention of the medical community and offers hope to thousands of families affected by similar inherited eye conditions.
Understanding the Rare Condition Behind Her Blindness
Leber congenital amaurosis affects approximately 1 in 40,000 newborns worldwide. This inherited disorder prevents the retina from properly detecting and processing light, resulting in severe visual impairment or complete blindness from birth.
Key characteristics of LCA include:
- Severe vision loss present at birth or within the first few months of life
- Abnormal eye movements known as nystagmus
- Poor pupillary light response
- Progressive retinal degeneration over time
- Occasional photophobia or sensitivity to light
In this young patient’s case, the condition was linked to a specific mutation in the RPE65 gene, which provides instructions for producing an enzyme essential for the visual cycle. Without functional RPE65 protein, the photoreceptor cells in the retina cannot respond to light effectively, leading to profound vision loss.
How Gene Therapy Works for Inherited Eye Disease
The gene therapy procedure administered to this 6-year-old represents one of the most targeted approaches in modern ophthalmology. The treatment, known commercially as voretigene neparvovec-rzyl (Luxturna), was specifically designed for patients with confirmed RPE65 mutation.
The procedure involves three critical steps:
- A subretinal injection — under general anesthesia, surgeons deliver a harmless viral vector carrying a functional copy of the RPE65 gene directly beneath the retina
- Cellular uptake — the viral vector penetrates retinal cells and delivers the therapeutic gene into their nuclei
- Protein production — once inside, the cells begin producing the missing enzyme, restoring the visual cycle
Dr. Sarah Chen, a pediatric ophthalmologist not involved in the case, notes:
“This isn’t a cure for all forms of blindness, but for carefully selected patients with RPE65-related LCA, gene therapy can produce dramatic, sometimes life-altering results. The therapy essentially rewires the retina to function as nature intended.”
The Before-and-After: A Parent’s Perspective
The girl’s parents described life before treatment as navigating a world of shadows and whispers. Their daughter relied heavily on tactile input and sound to understand her environment, unable to recognize faces or respond to visual cues that most children take for granted.
Life After Therapy: The First Signs of Sight
Within weeks of receiving the gene therapy, the family began noticing subtle but unmistakable changes. The once aimless eye movements grew more purposeful. She started tracking moving objects with her gaze and reaching for toys placed within her visual field.
Notable improvements documented by the family and medical team:
- Visual acuity improvement — from light perception only to measurable form and object recognition
- Navigation ability — ability to walk independently through unfamiliar spaces without bumping into obstacles
- Facial recognition — for the first time, the girl could identify her parents’ faces from across a room
- Environmental awareness — responsive to changes in lighting and movement in her peripheral vision
“Every day brings a new discovery,” the mother shared with reporters. “She points to birds in the sky now. She waves at people from a distance. These are moments we never dared to hope for.”
Why This Case Matters for the Future of Gene Therapy
While gene therapy for RPE65-related retinal disease has been FDA-approved since 2017, cases like this one continue to provide invaluable data about long-term outcomes and optimal treatment windows.
Pediatric Considerations in Gene Therapy
Children present unique challenges for gene therapy treatment. Their eyes are still developing, which requires specialized surgical techniques to avoid damaging delicate ocular structures. However, younger patients may also be better candidates because retinal degeneration has not progressed as far, potentially allowing for more complete visual restoration.
Critical factors influencing gene therapy success in children include:
- Age at treatment — younger patients tend to have healthier retinal cells to work with
- Genetic confirmation — only patients with the specific RPE65 mutation are eligible
- Baseline retinal structure — preserved photoreceptor density improves outcomes
- Surgical precision — the subretinal injection must be placed with millimeter accuracy
For this 6-year-old, the timing was optimal. Her retinal structure remained viable enough to respond to the therapy, yet she was old enough to cooperate with post-operative evaluations and vision rehabilitation.
The Emotional and Practical Impact of Restored Vision
The psychological transformation accompanying restored vision can be as significant as the physical changes. Children born with blindness develop coping mechanisms and neuroplastic adaptations that must integrate with new visual input.
Common emotional milestones include:
- Initial confusion or overwhelm from processing visual information
- Gradual curiosity and excitement about new visual experiences
- Joyful recognition of loved ones’ faces for the first time
- Increased confidence and independence in daily activities
Educational specialists emphasize that vision restoration requires patience. The brain must learn to interpret visual signals it has never processed before, which means occupational therapy, low-vision rehabilitation, and family support are essential components of the post-treatment journey.
Broader Implications for Rare Disease Treatment
This success story extends beyond ophthalmology into the wider field of rare genetic disease management. Gene therapies that once seemed theoretical are now producing real-world results, changing how we think about previously untreatable conditions.
Similar gene therapy approaches are being explored for:
- Other forms of inherited retinal degeneration, such as choroideremia and Stargardt disease
- Metabolic disorders like spinal muscular atrophy and certain enzyme deficiencies
- Blood disorders including hemophilia and beta-thalassemia
- Certain neurodegenerative conditions where delivery mechanisms can reach target tissues
This 6-year-old girl’s journey from darkness to light serves as a powerful reminder that genetic technology, when carefully applied to the right patients at the right time, can rewrite medical possibilities.
A Future Bright with Possibility
As gene therapy techniques improve and become more accessible, cases like this one will hopefully become less exceptional and more routine. For now, the image of a little girl seeing her parents’ faces for the first time stands as a testament to human ingenuity, medical perseverance, and the profound impact of restoring something as fundamental as sight.
The road ahead includes continued monitoring of long-term outcomes, refinements in surgical technique, and expanded eligibility criteria for more patients. But for one 6-year-old in particular, the world has already transformed from an abstract soundscape to a vivid, colorful landscape waiting to be explored.