Revolutionary Eye Implant Delivers 3 Years of Glaucoma Treatment
For the millions of people worldwide living with glaucoma, the daily routine of administering eye drops is a familiar, yet fraught, necessity. This “silent thief of sight” requires consistent, long-term pressure management to prevent irreversible vision loss. However, the very regimen designed to protect vision is often its own greatest hurdle. Now, a groundbreaking advancement from biomedical engineers offers a stunning solution: a tiny, refillable implant that can continuously deliver glaucoma medication for up to three years from a single procedure.
The Glaucoma Management Dilemma: A Problem of Consistency
Glaucoma is primarily characterized by increased intraocular pressure (IOP), which damages the optic nerve over time. The first line of treatment is almost always prescription eye drops. Yet, studies consistently show that patient adherence to drop regimens is notoriously poor, with rates often below 50%. The reasons are multifaceted:
- Complex Schedules: Multiple drops per day are difficult to remember.
- Physical Challenges: Arthritis, tremors, or poor vision make administering drops accurately a struggle.
- Side Effects: Stinging, redness, and blurred vision can discourage use.
- Simple Forgetfulness: Life gets in the way, even with the best intentions.
This inconsistency leads to dangerous peaks and troughs in IOP, leaving the optic nerve vulnerable to damage even when the patient believes they are being treated. The consequences can be devastating, progressing to tunnel vision and, ultimately, blindness.
Introducing the iDose TR: A Paradigm Shift in Treatment
Developed by Glaukos Corporation, the iDose TR is a novel therapeutic system that aims to replace the unreliable eye drop model with sustained, controlled drug delivery. This implant is a marvel of micro-engineering designed to seamlessly integrate with the eye’s natural anatomy.
How the Tiny Implant Works
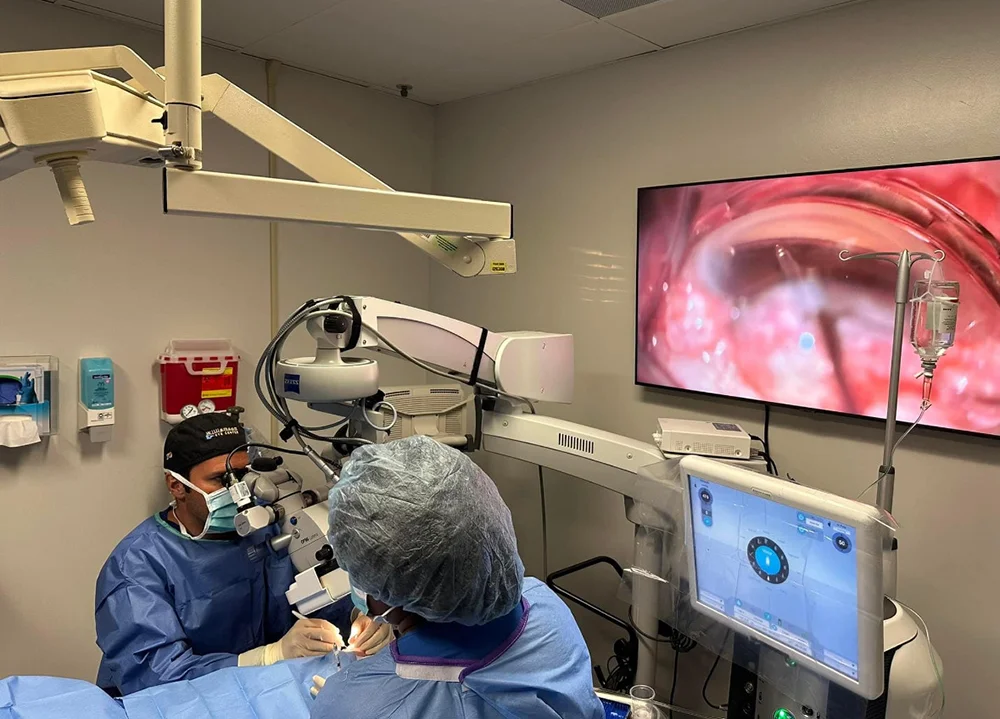
The iDose TR is a titanium implant, smaller than a grain of rice, that is surgically placed in the eye’s trabecular meshwork—the primary drainage area. The core of its innovation lies in its sealed reservoir pre-loaded with a proprietary formulation of Travoprost, a common and effective prostaglandin analog used to lower IOP.
The implant leverages the eye’s natural fluid dynamics. As aqueous humor flows past the implant, the drug elutes through a membrane at the tip at a precise, steady rate. This creates a continuous, 24/7 therapeutic effect, mimicking perfect patient adherence without any daily effort from the patient. Once the drug is depleted (after approximately three years), the empty implant can be removed and a new one inserted in a straightforward procedure, offering the potential for lifelong, maintenance-free management.
Beyond Adherence: The Multifold Advantages of Sustained Release
While solving the adherence crisis is the headline benefit, the advantages of a sustained-release implant like the iDose TR run much deeper.
- Elimination of Systemic Side Effects: Eye drops drain into the tear duct, where they are absorbed systemically and can sometimes cause side effects like low blood pressure or asthma exacerbation. The implant delivers medicine directly to the target site, minimizing systemic exposure.
- Reduced Ocular Surface Disease: Preservatives in eye drops, such as benzalkonium chloride (BAK), are known to damage the ocular surface, causing chronic dryness and inflammation. The implant’s preservative-free delivery protects the eye’s front surface.
- Superior IOP Control: Continuous delivery avoids the pressure spikes that can occur between drop doses, potentially offering more stable and effective IOP reduction over the long term.
- Life-Changing Convenience: The freedom from a daily burden cannot be overstated. It reduces anxiety, simplifies travel, and improves overall quality of life.
The Road to Clinical Practice: Evidence and Approval
The promise of the iDose TR is backed by robust clinical data. In pivotal Phase 3 trials, the implant demonstrated non-inferior IOP reduction compared to twice-daily Travoprost eye drops over the course of a year. Furthermore, it showed an excellent safety and tolerability profile.
Based on this compelling evidence, the iDose TR received approval from the U.S. Food and Drug Administration (FDA) in December 2023. This landmark approval paves the way for this technology to become a standard of care, offering ophthalmologists a powerful new tool in their fight against glaucoma progression.
Looking Ahead: The Future of Ophthalmic Implants
The iDose TR represents more than just a new device; it signifies a fundamental shift in how we approach chronic eye diseases. The success of this sustained-release model opens the door for a new era of bioengineering.
What This Means for Patients and Doctors
For patients, this technology is a beacon of hope. It transforms a manageable chronic condition from a daily struggle into a background concern addressed with a periodic, in-office procedure. It empowers individuals to live their lives without the constant reminder of their disease.
For ophthalmologists, it provides a reliable, effective, and practice-building procedure that addresses the core flaw in traditional glaucoma management. It allows them to ensure their patients are receiving the treatment they prescribe, leading to better clinical outcomes and preserved vision.
A Clearer Vision for Tomorrow
The development of the iDose TR implant is a triumph of biomedical innovation directly addressing a critical unmet need in global eye care. By decoupling effective treatment from daily patient action, it has the potential to prevent countless cases of vision loss attributed to poor adherence.
As this technology moves into broader clinical use and evolves further, it sets a new standard. The future of glaucoma treatment is no longer just about developing more potent drugs, but about creating intelligent delivery systems that ensure those drugs work as intended. This tiny implant is a giant leap toward a world where glaucoma’s progression can be reliably halted, allowing patients to preserve their sight and their quality of life for years to come.