Teprotumumab for Thyroid Eye Disease: What You Need to Know Now
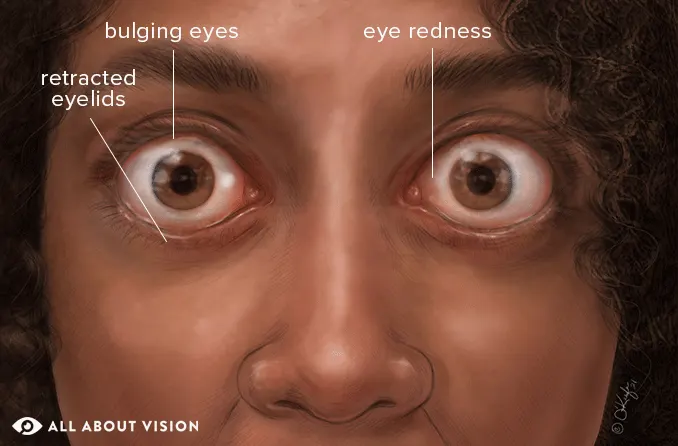
Thyroid Eye Disease (TED), also known as Graves’ ophthalmopathy, has long been one of the most difficult autoimmune eye conditions to treat. For years, management options were limited to steroids, radiation therapy, or surgery to control symptoms such as eye bulging, double vision, and orbital pain.
That changed with the introduction of teprotumumab (Tepezza®), the first FDA-approved drug designed to target the underlying biological process of TED rather than just its symptoms. It marked a major shift in treatment, but real-world experience has added important context to its use.
Today, clinicians emphasize both its promise and its limitations. This article breaks down how the drug works, what current evidence shows, and what patients should realistically expect.
How Teprotumumab Works in Thyroid Eye Disease
TED develops when the immune system mistakenly attacks tissues behind the eyes. A key driver is the activation of the insulin-like growth factor-1 receptor (IGF-1R) on orbital cells, which leads to inflammation and tissue expansion.
This process causes:
- Swelling of eye muscles and fat tissue
- Forward displacement of the eyes (proptosis)
- Double vision from muscle restriction
- Pressure and discomfort around the eyes
Teprotumumab is a monoclonal antibody that blocks IGF-1R activity. By interrupting this pathway, it reduces inflammation and prevents further tissue expansion. Over time, this can lead to visible improvement in eye appearance and function.
What Clinical Trials Showed vs. Real-World Experience
Early clinical trials, including the OPTIC study, showed strong results. Many patients experienced a meaningful reduction in eye bulging and improved quality of life compared to placebo.
However, real-world use has provided a more balanced picture:
- Some patients respond very well, with noticeable improvement in eye appearance and symptoms
- Others experience partial improvement only
- A smaller group shows little to no response
Experts now emphasize that results are not guaranteed, especially in more advanced or long-standing disease.
Timing Is Critical for Best Results
One of the most important findings in clinical practice is that timing strongly affects outcomes.
Teprotumumab works best during the active inflammatory phase of TED, when swelling and immune activity are still ongoing.
Ideal candidates typically include:
- Patients with recent-onset TED (often within 6–12 months)
- Moderate to severe active disease
- Evidence of ongoing inflammation
Less ideal candidates include:
- Patients with long-standing, stable disease
- Those with fibrotic or scarred orbital tissue
- Individuals whose symptoms have plateaued for years
Once fibrosis sets in, structural changes become harder to reverse, limiting the drug’s effectiveness.
Side Effects and Safety Considerations
While teprotumumab is targeted, it still carries important risks that require monitoring.
Common or clinically significant side effects include:
- High blood sugar (hyperglycemia): Especially in people with diabetes or prediabetes
- Hearing changes: Including tinnitus or hearing loss in some patients
- Muscle cramps and fatigue
- Infusion-related reactions: Headache, flushing, or mild respiratory symptoms
Because of these risks, patients typically need:
- Baseline blood sugar testing
- Hearing evaluation before and during treatment
- Ongoing monitoring throughout the full treatment course
This is an 8-infusion therapy given over several months, so consistent follow-up is essential.
How It Compares to Traditional Treatments
Before teprotumumab, treatment options were largely supportive or surgical.
- Steroids: Reduce inflammation but often temporary and systemic side effects are common
- Radiation therapy: Can help in some cases but has variable results
- Orbital decompression surgery: Effective for severe bulging but invasive and irreversible
Teprotumumab offers a more targeted approach by acting on the disease mechanism itself. For the right patient, it may reduce or delay the need for surgery.
However, it does not replace surgery in all cases, especially when structural changes are already permanent.
Who Should Consider Teprotumumab?
Based on current clinical guidance, good candidates generally include:
- Patients with active moderate-to-severe TED
- Early in the disease course
- No uncontrolled diabetes or significant pre-existing hearing loss
- Able to commit to intravenous treatment over several months
Patients with inactive disease or long-standing stable symptoms are less likely to benefit.
A specialist evaluation by an ophthalmologist experienced in orbital disease is essential before starting therapy.
Cost and Access Challenges
One of the biggest barriers is cost. A full treatment course can exceed $100,000 USD.
Although insurance coverage is often available for approved cases, patients may face:
- Prior authorization delays
- Coverage restrictions
- High out-of-pocket costs
Patient assistance programs may help reduce financial burden, but access can still be complex and time-consuming.
What the Future May Bring
Research into thyroid eye disease continues to evolve. Current areas of study include:
- Shorter or lower-dose treatment schedules
- Predictive markers for treatment response
- Combination therapies with other immune-targeting drugs
- Expanded use in earlier or more diverse patient groups
As more real-world data becomes available, treatment protocols are likely to be refined further.
Key Takeaways
Teprotumumab represents a major breakthrough in thyroid eye disease treatment, but it is not a universal solution.
What patients and clinicians should keep in mind:
- It works best in early, active disease
- Not all patients respond equally
- Side effects require monitoring, especially blood sugar and hearing
- Early specialist evaluation improves treatment success
- Cost and access remain significant barriers
While not perfect, teprotumumab has changed the treatment landscape for TED and offers meaningful improvement for many patients who previously had limited options.